May 16
Experiment – metal hotter or plastic?
The question was – if two lids are left overnight, one metal and one plastic, which one would be hotter? After carrying out the experiment, it was proved that they were both very similar, with a difference of 0.2 degrees. However, the question was that, if they were both the same, why does the metal feel colder.
The explanation is that since metal is a good conductor of heat, it can transfer heat and absorb heat from other objects. So, if a person were to touch the metal, the metal would ‘suck’ the heat/thermal energy from the fingers of the person much faster than the plastic would, since the plastic is the insulator. Hence, your hands would feel colder when you touch the metal as compared to when you touch the plastic.

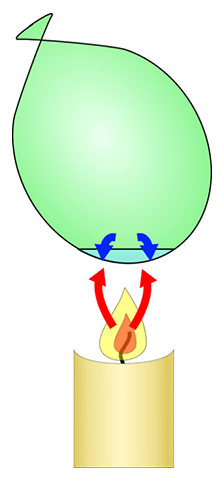
Experiment – water balloon vs balloon without water
The second experiment asked the question that if there were 2 balloons, one with a little bit of water, one without any water, and both of them were put over a bunsen burner, what would happen? What happened was that the one without the water popped much earlier than the one with the water.
The explanation for this is that in the one with the water, the water absorbs the heat from the bunsen burner, so the rubber of the balloon does not heat up and expand as fast, because the heat is being absorbed by the water. The ability of water to soak up and retain heat is really good. In the one without the water, there was no water to absorb the heat, so the rubber of the balloon expands and heats up faster, and hence it pops faster.
After unit reflection: In this post I analysed the lids and water balloons to apply the concepts of energy. By analyzing both the lid and the balloon I applied the concept of kinetic energy and temperature, as well as the concept of heat conduction. By explaining that since metal conducts heat faster, the molecules move around faster, and it can absorb and give heat faster, and how water absorbs heat, these I analyzed and explained these principles.